States of Matter
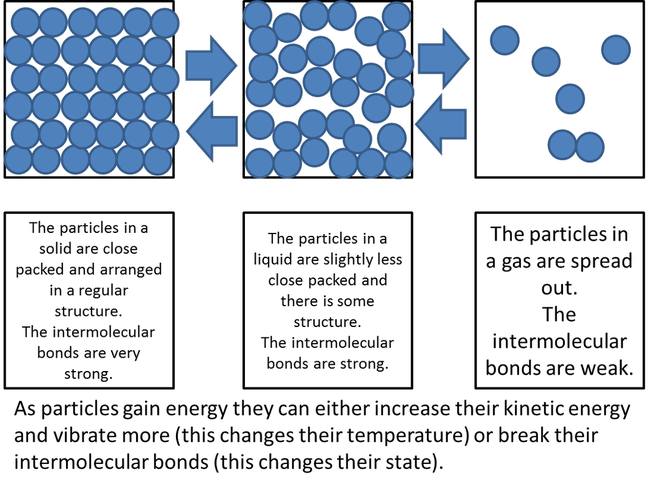
All matter is made up of particles. These particles can be arranged in three ways or three states: as a solid, a liquid, or a gas. These particles move around randomly, colliding with each other and with the walls of their container, and vibrate.
|
Remember, the transition from solids to liquids to gases is a continuous scale.
Pressure: Gases will exert pressure of the walls of their container. They do this by colliding with the walls as they move around randomly. As they rebound off the walls they exert a force. At higher temperatures they will be moving around faster and so hit the walls with a harder force and more often. |
|
|
Boiling and Evaporation
The boiling point of a substance is when all of the liquid turns into a gas. This happens at a particular temperature, the boiling point. Evaporation happens at the surface of a liquid all the time: there will be some particles that have enough energy to break free and become a gas. You can increase the rate of evaporation by having a larger surface area and a cool breeze. Impurities You can increase the boiling point of a liquid by adding impurities. You can decrease the melting point of a substance by adding impurities; that's why salt and grit are put on the roads in the winter! |
You may find the following Phet simulation useful: