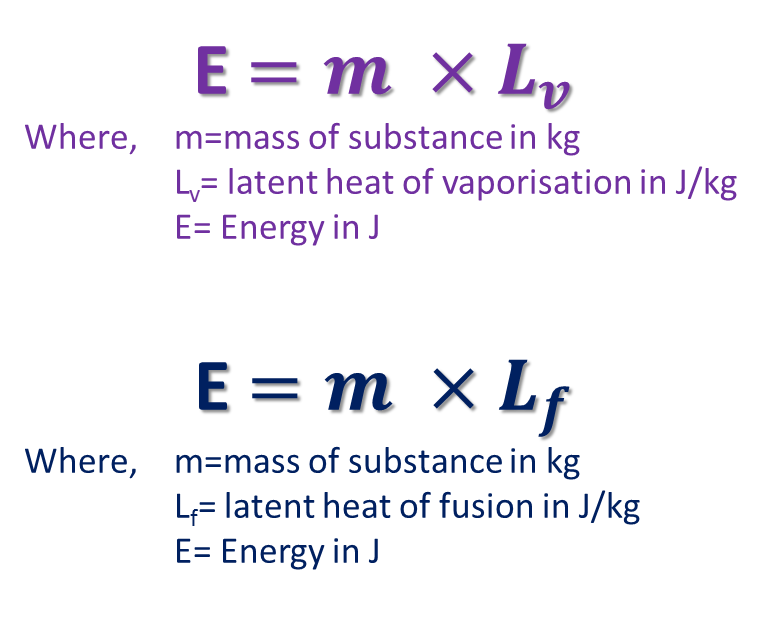Specific Latent Heat
Specific latent heat is the energy required to change the state of 1kg of a substance without changing its temperature. It is measured in J/kg
|
Specific latent heat of fusion is the energy required to change 1kg of a substance from a solid to a liquid without changing its temperature. This will happen at its melting point.
When a substance is melting, it will take in this energy; when it is freezing it will give out this energy. Specific latent heat of vaporisation is the energy required to change 1kg of a substance from a liquid to a gas without changing its temperature. This will happen at its boiling point. When a substance is boiling, it will take in this energy; when it is condensing it will give out this energy. This energy is used by the substance to break or make intermolecular bonds. Think of it as the energy stored in those bonds. Its name comes from the latin 'lateo', 'I lie hidden'. |
|
Keeping cool by evaporationHave you noticed that nail varnish remover feels cold when you spill it on your skin?
Why does sweat cool us down? It's all to do with energy being used to break the intermolecular bonds and allow a liquid to turn into a gas. This energy is the specific latent heat of vaporisation. Check out the video for an explanation! |
|
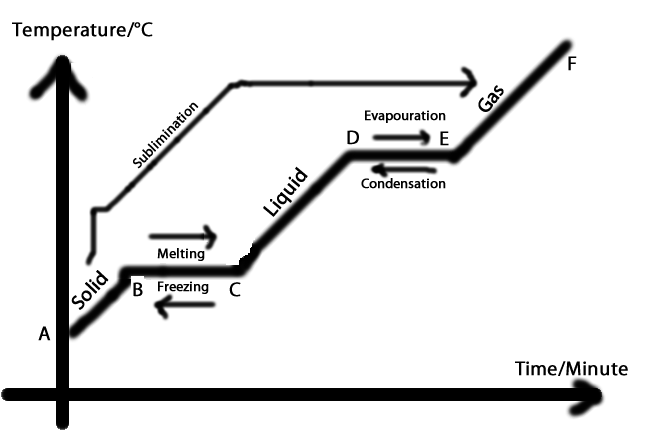
The graph shows a pure hydrocarbon being heated.
If the temperature is increasing the energy is being used to increase the kinetic energy of the molecules, i.e. to make them vibrate faster. If the temperature is constant, the energy is being used to break the intermolecular bonds, i.e. to make the molecules move further apart. They will still be moving at the same speed because they have the same temperature but they won't be as close together. This constant temperature will happen every time the substance changes state. The energy absorbed during that time will be equal to the latent heat. |