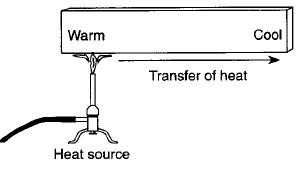
Conduction
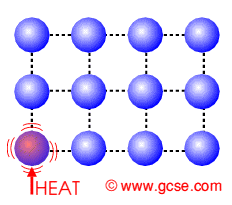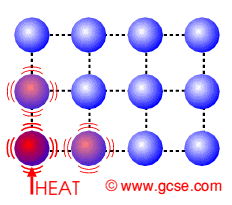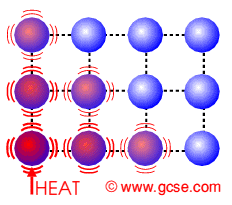
Conduction is how heat is transferred in solids. Particles near the source of heat gain more kinetic energy and vibrate more. They can pass this energy onto their neighbours.
|
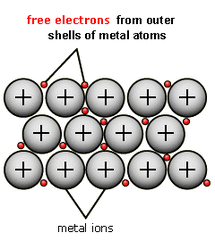
Conduction in Metals
Metals consist of a lattice of molecules in a regular structure and a sea of free electrons. This cloud of electrons can move around very easily. These electrons can pass on their heat energy to each other. |
Conduction in Insulators
Insulators do not have free electrons and so the heat causes the lattice of molecules to vibrate. They pass on their energy to neighbouring molecules. |
A substance that transfers heat energy well is called a conductor and one that transfers it poorly is called an insulator. Metals are good conductors.