Half Life
The half life of a radioactive substance is the time it takes for its activity level to decrease by half.
|
Half-life is a measure of how radioactive a substance is.
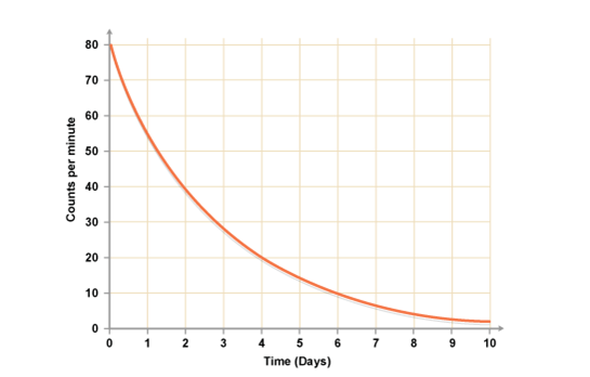
Once a nucleus has decayed it will become stable and not decay any further. This means that over time there will be fewer nuclei withing a radioactive substance that are able to decay. Because of this, its activity will drop. If we plot a graph of the activity against time of any radioactive sample it will exponentially decay like the graph on the right. We use this graph to calculate the half life: you can start at any point on the y axis and measure the time it takes to decrease by half (eg. 80 to 40 counts/min or 20-10 counts/min). The answer is always the same: two days. |
This means that every two days the activity will drop by 50%.
So after two days, one half life, the activity will be 50% of its starting activity. After four days, two half lives, it will be 25% of its starting activity. After six days, three half lives, it will be 12.5% of its starting activity. |
Carbon Dating
Half life is used to find out how old things are by a process called carbon dating. Have a look at this Bang Goes The Theory Clip to find out more.