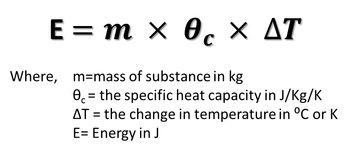Specific Heat Capacity
Specific Heat Capacity is the energy required to change the temperature of 1kg of a substance by 1 degrees Celsius or Kelvin
Definition:If the temperature of a substance is changing then this is how to calculate the energies involved.
If a substance is cooling, this will be the energy released to the surroundings. If a substance is heating up, this is the energy you have to give it. You can also think of it as the energy stored in a substance. A substance with a higher specific heat capacity will store more energy. This is the vibrational energy of the molecule. |