Specific Heat Capacity Experiment
Aim: to calculate the specific heat capacity of lead and aluminium
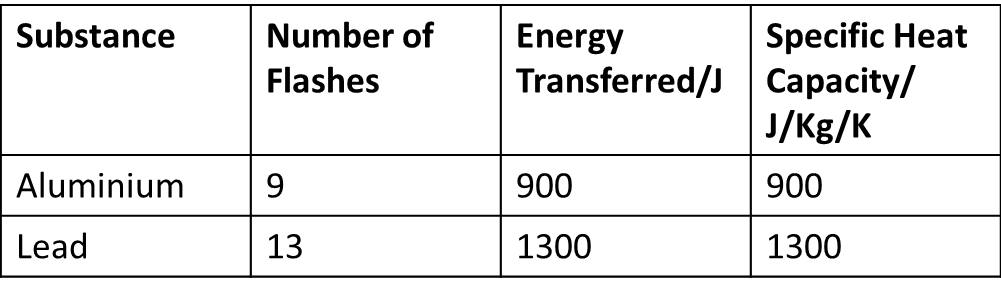
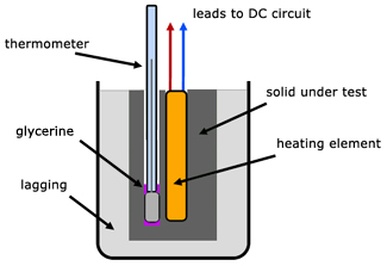
Hypothesis: I think the specific heat capacity of lead will be higher than that of aluminium. Method: 1) Place a Joule heater in the 1Kg lead block and connect it to a power supply 2) Put a little bit of glycerol on the thermometer (this makes the temperature reading more accurate) and place it in the 1Kg lead block. 3) Switch the power supply on and count the number of flashes that occur while the temperature changes by 1 degrees C. 4) Each flash on the Joulemeter is 100J. 5) You can calculate the specific heat capacity: specific heat capacity = Energy/ (mass x temperature change) 6) Repeat with Aluminium block. Results: Conclusion:
The specific heat capacity of Alumium is 900J/Kg/K and the specific heat capacity of Lead is 1300J/Kg/K. This is as I predicted. This means that it takes more energy to change the temperature of lead than of Aluminium. ExtensionHave a look at a different way of calculating the specific
heat capacity. This way they are using the a hot metal to heat water and looking at the temperture change. We know that the heat energy from the metal MUST have been transfered to the water. |
Evaluation:
The precision of the thermometer was to the nearest 0.5 degrees. That is a large error if we are only looking for a 1 degree change in temperature. We could reduce the error in the experiment by heating the block up by 10 degrees rather than just by 1. This will make our results more accurate. The accuracy of the flashes depends on the power supplied to the Joulemeter. We could improve the experiment by using a voltmeter, ammeter and stopwatch and measuring the energy transfered directly by using E=VIt. This would make our reading more accurate. Another problem with the experiment was heat loss to the surroundings through the block. We could reduce heat loss by insulating the metal block or by calculating the rate of cooling and applying that factor. This would make our result more accurate. We could improve the reliability of the experiment by taking more repeats and calculating an average. At the end the water and the metal are at equilibrium tempertures (the same). We know the specific heat capacity of water is 4200J/Kg/K. The energy transfered to the water can be calculated using: Energy =mass x specific heat capacity x temperature change This will be the same energy change as the metal block. The specific heat capacity can then be calculated using: Specific heat capacity = energy/(mass x temperature change) Can you work it out? Remember: the temperature changes for the metal block and the water are different! |