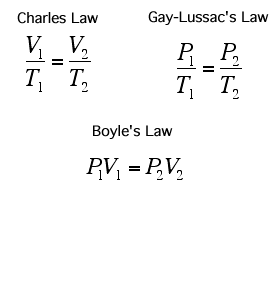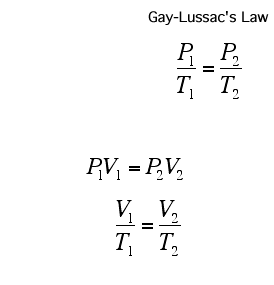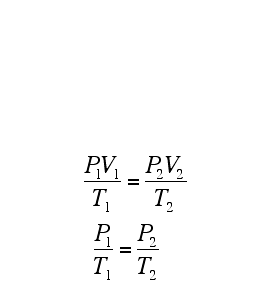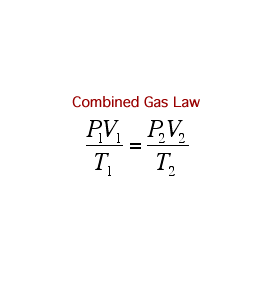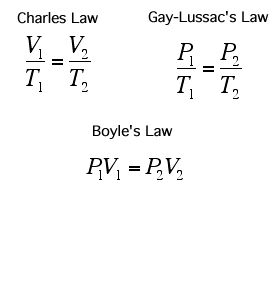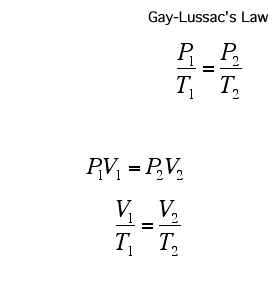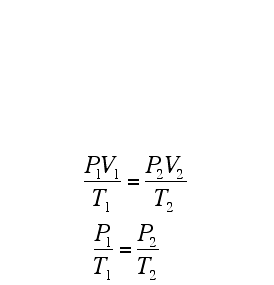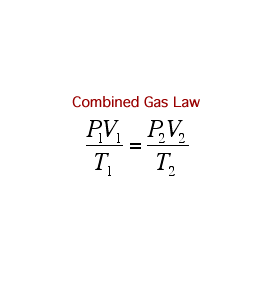Ideal Gases
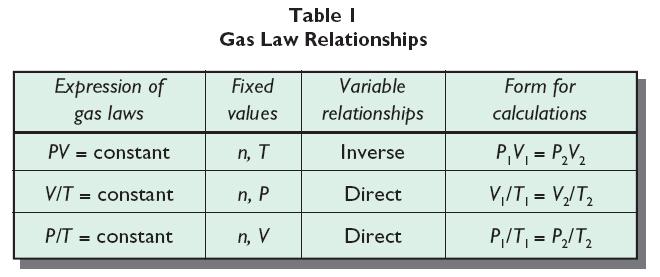
Pressure in a Gas
The pressure in a gas comes from the random motion of its particles and their collisions with the walls of its container: the pressure can be increased by more frequent or harder collisions.
The pressure in a gas is proportional to its temperature: if we increase the temperature of a gas, we increase the average kinetic energy of the gas molecules. This means that they are moving around faster and so are hitting the walls more often and harder, at a higher speed, thus increasing the pressure.
We can also increase the pressure by increasing the number of particles in the gas, so increasing the concentration.
The pressure in a gas will also increase it the volume of the gas decreases. This is because it will hit the walls of the container more frequently.
The pressure in a gas is proportional to its temperature: if we increase the temperature of a gas, we increase the average kinetic energy of the gas molecules. This means that they are moving around faster and so are hitting the walls more often and harder, at a higher speed, thus increasing the pressure.
We can also increase the pressure by increasing the number of particles in the gas, so increasing the concentration.
The pressure in a gas will also increase it the volume of the gas decreases. This is because it will hit the walls of the container more frequently.
Temperature of a Gas (Charle's Law)
If you increase the temperature of a gas, it will want to expand and increase its volume. This is because as the temperature gets higher, the particles gain more kinetic energy and will move around more, this will hit the sides of the container more frequently and with a greater force.
Remember, if you are using the formulae above, the temperature has to be in Kelvin!
You might find the following simulation useful:
|
|
|