Specific Latent Heat of Fusion Experiment
|
Aim: to investigate how the temperature changes with time as a hydrocarbon cools.
Hypothesis: I think my hydrocarbon will cool steadily. Method: 1) Heat up a test tube of the hydrocarbon in a water bath until it reaches 80 degrees. 2) Take the test tube out of the water bath and record the temperature every 30s for the next 10 minutes using a stop watch and a thermometer. 3) Heat the hydrocarbon back up to 80 degrees in the water bath and repeat the experiment. |
Results:
Conclusion:
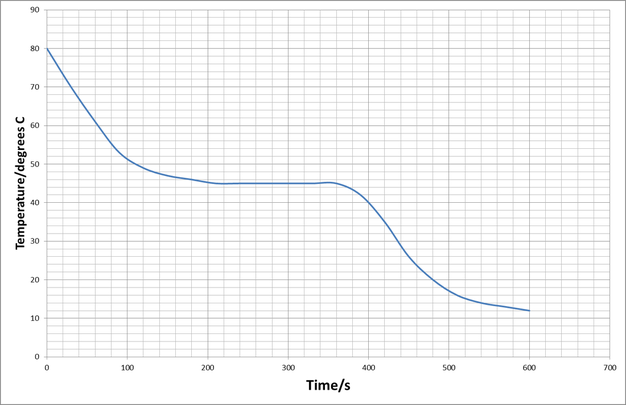
At first the temperature decreases. The rate of temperature change decreases as the liquid cools. At 200s the temperature stays at a constant temperature of 45 degrees C until 320s. The temperature continues to decrease after that, again with the rate of cooling decreasing with time.
The rate of cooling decreases with temperature. This is because the rate of cooling will depend on the temperature difference between the substance and its surroundings.
The area of constant temperature is while the hydrocarbon is changing state.
At first the substance is losing energy as a drop in temperature: at this point the molecules are losing kinetic or vibrational energy.
At 200s the substance changes state; at this point it is losing the energy that keeps the molecules apart, the energy that can break the intermolecular bonds. This will make the molecules in the substance move closer together and the substance will change from a liquid to a solid.
The substance then continues to lose energy as vibration energy and so the temperature decreases.
At first the temperature decreases. The rate of temperature change decreases as the liquid cools. At 200s the temperature stays at a constant temperature of 45 degrees C until 320s. The temperature continues to decrease after that, again with the rate of cooling decreasing with time.
The rate of cooling decreases with temperature. This is because the rate of cooling will depend on the temperature difference between the substance and its surroundings.
The area of constant temperature is while the hydrocarbon is changing state.
At first the substance is losing energy as a drop in temperature: at this point the molecules are losing kinetic or vibrational energy.
At 200s the substance changes state; at this point it is losing the energy that keeps the molecules apart, the energy that can break the intermolecular bonds. This will make the molecules in the substance move closer together and the substance will change from a liquid to a solid.
The substance then continues to lose energy as vibration energy and so the temperature decreases.
|
Evaluation:
The precision of my theremometer was to the nearest 0.5 degrees C and it was quite difficult to measure without a paralax error. I could improve this by using a digital thermometer, which would make my results more accurate. The precision of the stop watch was to the nearest 0.01s but with reaction time in reading the thermometer the timings were probably only accurate to the nearest 5s. I would improve this by attaching a data logger to my thermometer. This could take continuous readings, making my results more reliable, and could plot a graph automatically. This would allow me to measure how long the hydrocarbon took to change state more accurately. |
Still a bit confused? Have a look at the video above.
It explains what specific latent heat is and then has a different experiment. This practical is one that can measure the specific latent heat of fusion for water rather than just observing how it cools. |