Boiling point of water experiment
|
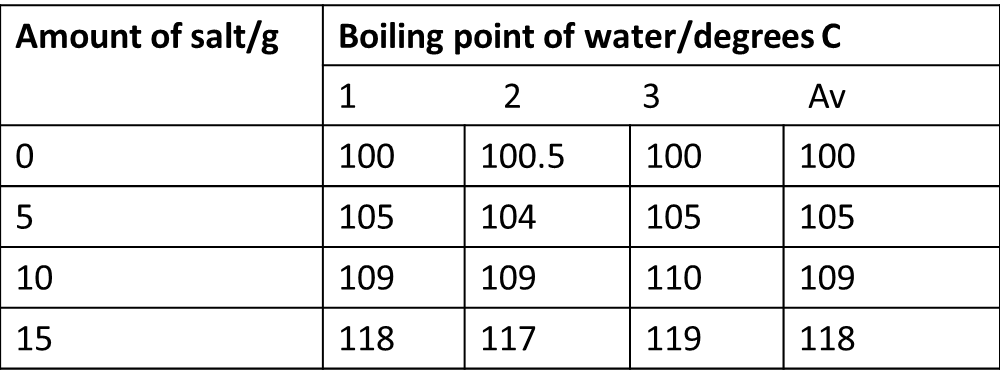
Aim: to investigate the relationship between the amount of salt in a beaker of water and the boiling point of the solution.
Hypothesis: I think that salt will incease the boiling point of water. Method: Add 5g of salt to 200ml of water and heat it with a bunsen burner. Record the temperature at which the water boils. Repeat the experiment with 0g, 5g, 10g, 15g of salt, testing each one three times. Results: Conclusion: my results show that salt increases the boiling point of water. For example with no salt the water boiled at 100 degrees C but with 15g of salt the water boiled at 118 degrees.
Evaluation: My repeat results were within 1 degree C of the average. This is quite reliable, however the accuracy in reading the thermometer was to the nearest 0.5 degrees C so other factors must have affected this. These factors could be the fact that the precision of the mass balance was only 0.1g or that the thermometer might not have had time to settle at a temperature. We could improve this experiment by using a digital thermometer, making the temperature reading more accurate and increasing the temperature of the water more slowly so that the thermometer could settle at the temperatures. |